Precipitate Reactions
Precipitation is the formation of a solid in a solution or inside another solid during a chemical reaction or by diffusion in a solid. When the reaction occurs in a liquid, the solid formed is called the Precipitate.
During chemical reactions, precipitation may also occur particularly if an insoluble substance is introduced into a solution
and the density happens to be greater (otherwise the precipitate would float or form a suspension). With soluble substances,
precipitation is accelerated once the solution becomes supersaturated.
During chemical reactions, precipitation may also occur particularly if an insoluble substance is introduced into a solution
and the density happens to be greater (otherwise the precipitate would float or form a suspension). With soluble substances,
precipitation is accelerated once the solution becomes supersaturated.
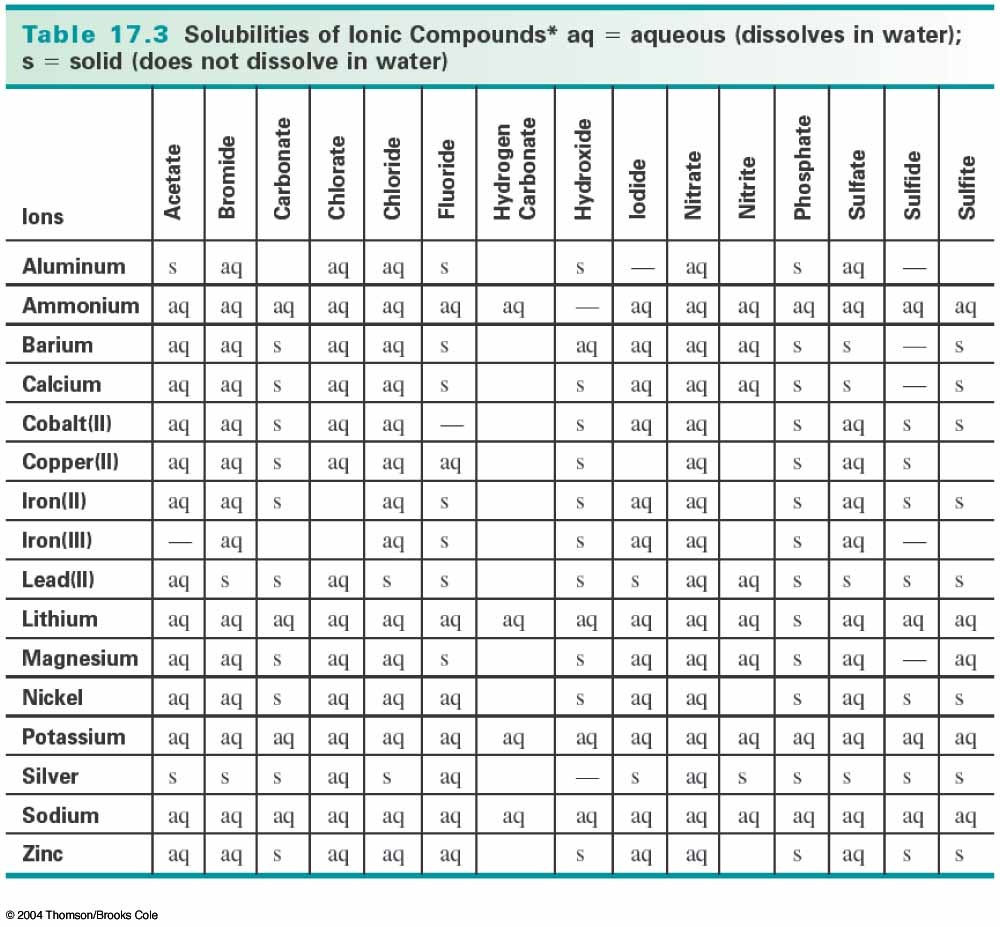
When two aqueous solutions of ionic compounds are mixed together, the resulting reaction may produce a solid precipitate. This guide will show how to use the solubility chart and rules to predict whether or not the product will remain in solution or form a precipitate.
When two aqueous solutions are mixed, the ions interact to form products.
AB(aq) + CD(aq) --> Products
This is generally a Double Displacement reaction.
AB(aq) + CD(aq) --> AD + CB
A precipitate will form if the resulting compound is insoluble in water. For example, a silver nitrate solution (AgNO3) is mixed with a solution of magnesium bromide (MgBr2). The balanced reaction would be:
2 AgNO3(aq) + MgBr2 --> 2 AgBr(?) + Mg(NO3)2(?)
The state of the products needs to be determined. Are the products soluble in water?
According to the solubility rules, all silver salts are insoluble in water with the exception of silver nitrate, silver acetate and silver sulfate. Therefore, AgBr will precipitate out.
The other compound Mg(NO3)2 will remain in solution because all nitrates, (NO3)-, are soluble in water. The resulting balanced reaction would be:
2 AgNO3(aq) + MgBr2 --> 2 AgBr(s) + Mg(NO3)2(aq)
The solubility Chart and Rules will help you predict the precipitate from the reaction.
Reference 3
When two aqueous solutions are mixed, the ions interact to form products.
AB(aq) + CD(aq) --> Products
This is generally a Double Displacement reaction.
AB(aq) + CD(aq) --> AD + CB
A precipitate will form if the resulting compound is insoluble in water. For example, a silver nitrate solution (AgNO3) is mixed with a solution of magnesium bromide (MgBr2). The balanced reaction would be:
2 AgNO3(aq) + MgBr2 --> 2 AgBr(?) + Mg(NO3)2(?)
The state of the products needs to be determined. Are the products soluble in water?
According to the solubility rules, all silver salts are insoluble in water with the exception of silver nitrate, silver acetate and silver sulfate. Therefore, AgBr will precipitate out.
The other compound Mg(NO3)2 will remain in solution because all nitrates, (NO3)-, are soluble in water. The resulting balanced reaction would be:
2 AgNO3(aq) + MgBr2 --> 2 AgBr(s) + Mg(NO3)2(aq)
The solubility Chart and Rules will help you predict the precipitate from the reaction.
Reference 3
|
Solubility Rules:
|
Example

The liquid in the photo has barium ions (Ba2+) in it. The liquid being added has sulfate ions (SO42-). When these mix, the barium ions combine with the sulfate ions to form solid barium sulfate (BaSO4).
Reference 1
Reference 1
Activities and Review Sheets
References
1) "Solubility Rules." About.com Chemistry. N.p., n.d. Web. 15 Oct. 2012. <http://chemistry.about.com/od/solutionsmixtures/a/solubility-rules.htm>.
2) "How to Predict Precipitates." About.com Chemistry. N.p., n.d. Web. 15 Oct. 2012. <http://chemistry.about.com/od/workedchemistryproblems/a/How-To-Predict-Precipitates.htm>.
2) "How to Predict Precipitates." About.com Chemistry. N.p., n.d. Web. 15 Oct. 2012. <http://chemistry.about.com/od/workedchemistryproblems/a/How-To-Predict-Precipitates.htm>.