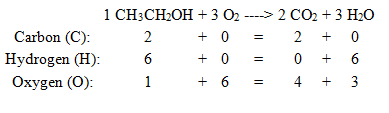Balancing chemical equations
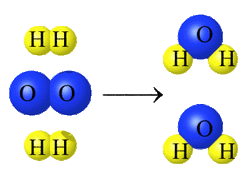
In a chemical reaction, atoms are NOT created or destroyed. What changes in a chemical reaction is the connections (bonds) between atoms. In the first reaction shown above, 2 molecules of two hydrogen atoms (H) start out bonded to each other.
During the course of the chemical reaction, the 2 H-H bonds break and 2 new H2O bonds ar3 formed.
During the course of the chemical reaction, the 2 H-H bonds break and 2 new H2O bonds ar3 formed.
How to?
1) Be sure that the formulas for each compound are written correctly. (Usually this will be taken care of).
2) Select the "simplest" element to begin balancing the equation. The "simplest" element is an element that only
occurs in one compound in the reactants and in one compound in the products.
3) Change the coefficients of the compounds containing the selected element so that the same number of atoms
of this element occur on both the reactant and product sides of the equation.
4) Go back to Step #2, selecting the "next simplest" element. Repeat steps 2-4 as until every element is "balanced".
5) After you are finished, check the final balanced equation to make sure every element is still balanced.
*** If you change a coefficient a second time, you will probably cause one of the 'earlier' elements to become 'unbalanced'.***
Example of the Combustion of Ethanol:
CH3CH2OH + O2 --> CO2 + H2O
In this reaction, all of the compounds have the correct formulas. The next step is to select the 'simplest' element. Either carbon (C) or hydrogen (H) could be used. For this example, we will select carbon.
Following step #3, we change the coefficients in front of ethanol (CH3CH2OH) and carbon dioxide (CO2). Since
there are two carbon atoms in ethanol and only one carbon atom in CO2, we add a two in front of CO2.
1 CH3CH2OH + __O2 --> 2 CO2 + __H2O
This equation now has two carbon atoms on the reactant side and two carbon atoms on the product side, so the carbon atoms are balanced. The "next simplest element" is hydrogen (H). Like before, we change the coefficients
in front of ethanol and water (H2O) to balance this element. One ethanol molecule contains six (6) hydrogen atoms, and one water molecule contains two (2) hydrogen atoms. To balance this, we need three water molecules (3x2=6).
1 CH3CH2OH + __O2 --> 2 CO2 + 3 H2O
Finally, we need to balance the oxygen atoms. On the reactants (left) side of this equation we have three (3) oxygen atoms, while on the product (right) side we have seven (7) oxygen atoms. (Two O atoms/CO2 times 2 = 4 oxygen atoms
PLUS one O atom/H2O times 3 = 3 oxygen atoms. 4+3=7).
Since the reactant side has fewer oxygen atoms, we need to change one or both coefficients to increase the number of oxygen atoms. However, we do NOT want to change the coefficient in front of ethanol because we have
already used this to balance carbon. (This is why I put a one in front of this
molecule). Changing the coefficient in front of oxygen (O2) will give the correct answer.
1 CH3CH2OH + 3 O2 --> 2 CO2 + 3 H2O
Finally, it is a good idea to check to be sure the equation really is still balanced.
(See Reference 1)
2) Select the "simplest" element to begin balancing the equation. The "simplest" element is an element that only
occurs in one compound in the reactants and in one compound in the products.
3) Change the coefficients of the compounds containing the selected element so that the same number of atoms
of this element occur on both the reactant and product sides of the equation.
4) Go back to Step #2, selecting the "next simplest" element. Repeat steps 2-4 as until every element is "balanced".
5) After you are finished, check the final balanced equation to make sure every element is still balanced.
*** If you change a coefficient a second time, you will probably cause one of the 'earlier' elements to become 'unbalanced'.***
Example of the Combustion of Ethanol:
CH3CH2OH + O2 --> CO2 + H2O
In this reaction, all of the compounds have the correct formulas. The next step is to select the 'simplest' element. Either carbon (C) or hydrogen (H) could be used. For this example, we will select carbon.
Following step #3, we change the coefficients in front of ethanol (CH3CH2OH) and carbon dioxide (CO2). Since
there are two carbon atoms in ethanol and only one carbon atom in CO2, we add a two in front of CO2.
1 CH3CH2OH + __O2 --> 2 CO2 + __H2O
This equation now has two carbon atoms on the reactant side and two carbon atoms on the product side, so the carbon atoms are balanced. The "next simplest element" is hydrogen (H). Like before, we change the coefficients
in front of ethanol and water (H2O) to balance this element. One ethanol molecule contains six (6) hydrogen atoms, and one water molecule contains two (2) hydrogen atoms. To balance this, we need three water molecules (3x2=6).
1 CH3CH2OH + __O2 --> 2 CO2 + 3 H2O
Finally, we need to balance the oxygen atoms. On the reactants (left) side of this equation we have three (3) oxygen atoms, while on the product (right) side we have seven (7) oxygen atoms. (Two O atoms/CO2 times 2 = 4 oxygen atoms
PLUS one O atom/H2O times 3 = 3 oxygen atoms. 4+3=7).
Since the reactant side has fewer oxygen atoms, we need to change one or both coefficients to increase the number of oxygen atoms. However, we do NOT want to change the coefficient in front of ethanol because we have
already used this to balance carbon. (This is why I put a one in front of this
molecule). Changing the coefficient in front of oxygen (O2) will give the correct answer.
1 CH3CH2OH + 3 O2 --> 2 CO2 + 3 H2O
Finally, it is a good idea to check to be sure the equation really is still balanced.
(See Reference 1)
Ionic vs molecular reaction
Ionic: The general statement of a chemical which shows only those substances actually undergoing change; thus Ba++ + SO4-- = BaSO4.
Molecular: the specific statement of a chemical reaction showing the relative proportions o substances involved;
thus: BaCl2 + H2SO4 = BaSO4 + 2HCl.
(See Reference 2)
Molecular: the specific statement of a chemical reaction showing the relative proportions o substances involved;
thus: BaCl2 + H2SO4 = BaSO4 + 2HCl.
(See Reference 2)
REferences
1) "Balancing Chemical Equations." Balancing Chemical Equations. N.p., n.d. Web. 15 Oct. 2012. <http://www.personal.kent.edu/~cearley/ChemWrld/balance/balance.htm>.
2) Hackh, Ingo Waldemar Dagobert, and Julius Grant. Hackh's Chemical Dictionary. USA: McGraw-Hill, Inc, 1969. Print.
2) Hackh, Ingo Waldemar Dagobert, and Julius Grant. Hackh's Chemical Dictionary. USA: McGraw-Hill, Inc, 1969. Print.